New publication in eLife of the Jordan Lab in collaboration with the IBMB Imaging Platform showing that…
A novel target to treat aggressive lymphomas is discovered by the Liesa laboratory in collaboration with Bantam Pharmaceutical
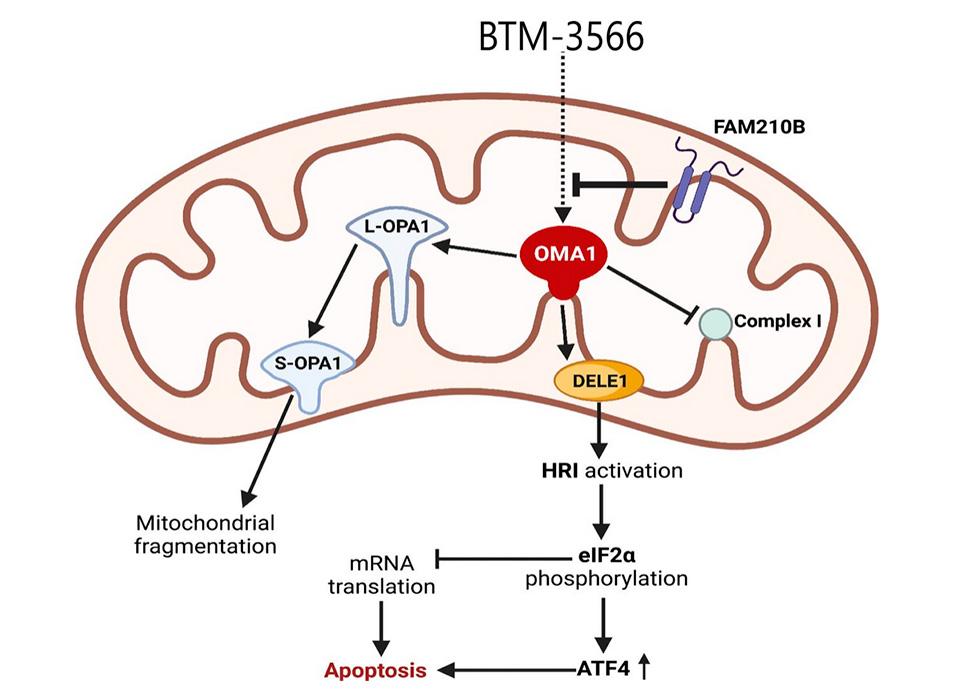
Dr. Liesa’s laboratory identified that the activation of the mitochondrial protease OMA1 is central to the action of a novel drug (BTM 3566) that cause complete tumor regression across a panel of Diffuse-Large B-Cell Lymphomas (DLBCL) patient-derived xenografts. The study was published in Molecular Cancer Therapeutics, from the American Association of Cancer Research (AACR).
Abstract
DLBCL are aggressive, rapidly proliferating tumors that critically depend on the ATF4-mediated integrated stress response (ISR) to adapt to stress caused by uncontrolled growth, such as hypoxia, amino acid deprivation and accumulation of misfolded proteins. Here we show that ISR hyperactivation is a targetable liability in DLBCL. We describe a novel class of compounds represented by BTM-3528 and BTM-3566, that activate the ISR through the mitochondrial protease OMA1. Treatment of tumor cells with compound leads to OMA1-dependent cleavage of DELE1 and OPA1, mitochondrial fragmentation, activation of the eIF2α-kinase HRI, cell growth arrest and apoptosis. Activation of OMA1 by BTM-3528 and BTM-3566 is mechanistically distinct from inhibitors of mitochondrial electron transport, as the compounds induce OMA1 activity in the absence of acute changes in respiration. We further identify the mitochondrial protein FAM210B as a negative regulator of BTM-3528 and BTM-3566 activity. Overexpression of FAM210B prevents both OMA1 activation and apoptosis. Notably, FAM210B expression is nearly absent in healthy germinal-center B-lymphocytes and in derived B-cell malignancies, revealing a fundamental molecular vulnerability which is targeted by BTM compounds. Both compounds induce rapid apoptosis across diverse DLBCL lines derived from activated B-cell, germinal center B-cell, and MYC-rearranged lymphomas. Once-daily oral dosing of BTM-3566 resulted in complete regression of xenografted human DLBCL SU-DHL-10 cells and complete regression in 6 of 9 DLBCL patient-derived xenografts. BTM-3566 represents a first- of-its kind approach of selectively hyperactivating the mitochondrial ISR for treating DLBCL.
Reference:
Adrian Schwarzer, Matheus Oliveira, Marc-Jens Kleppa, Scott D. Slattery, Andy Anantha, Alan Cooper, Mark Hannink, Axel Schambach, Anneke Doerrie, Alexey Kotylarov, Matthias Gaestel, Todd Hembrough, Jedd Levine, Michael Luther, Michael Stocum, Linsey Stiles, David M. Weinstock, Marc Liesa*, Matthew J. Kostura*; Targeting aggressive B-cell lymphomas through pharmacological activation of the mitochondrial protease OMA1. Mol Cancer Ther 2023; https://doi.org/10.1158/1535-7163.MCT-22-0718
*Marc Liesa and Matthew J. Kostura contributted equally to the article and are co-corresponding authors.