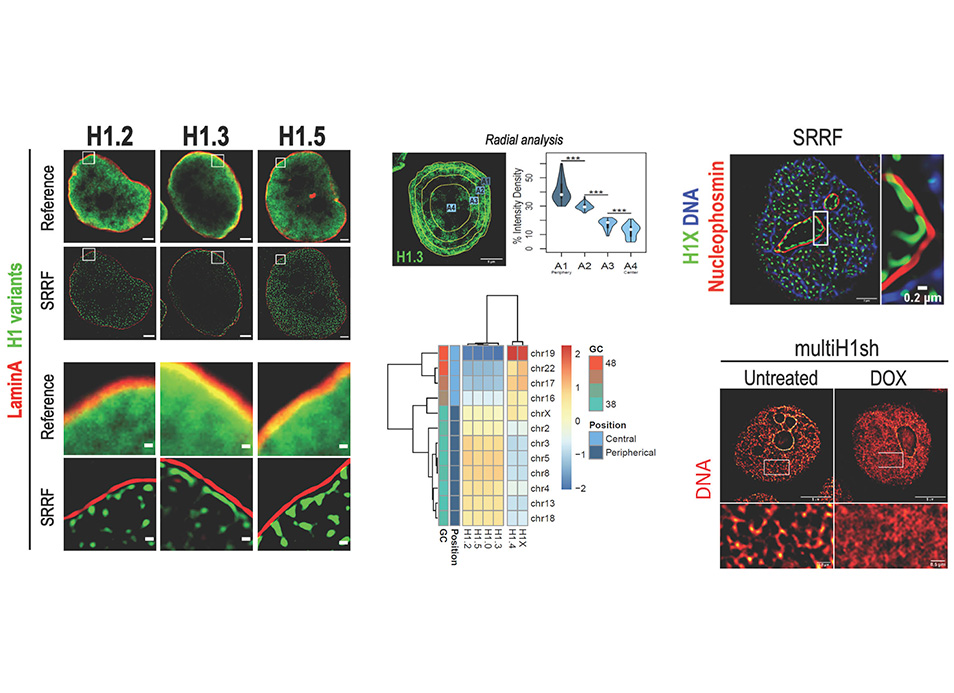
New publication in eLife of the Jordan Lab in collaboration with the IBMB Imaging Platform showing that…
“Multi-antibiotic resistance will be a serious medical problem for the future”
Laia Vives (04/04/2018)
Anna Rubio was born in Barcelona. She is doctor in Biotechnology and she went to Heidelberg (Germany) to do a postdoc four years ago. She recently published a study on bacterial transfer of antibiotic resistance at Cell journal. She is passionate about African dance and mountains and she loves travelling. She is giving the SCB Programme Seminar at Parc Científic de Barcelona tomorrow April 11th at 9:30am.
How did you begin your science career?
I studied Biochemistry and Biotechnology at the Autonomous University of Barcelona and then I did a master’s degree in Molecular Biotechnology at the University of Barcelona. The master’s degree led me to do the PhD at Dr. Maria Solà group at the Structural Biology Unit from Institute of Molecular Biology of Barcelona (Spanish Research Council).
The results from your PhD were published very well.
I was fortunate to take part of a fascinating research project about a human mitochondrial transcription factor (TFAM) directed by Dr. Maria Solà. We obtained its three-dimensional structure using X-ray crystallography. The structure and various biochemical assays allowed us to understand how mitochondrial DNA is packaged and how this factor can bend DNA and recruit the transcription machinery to express the genetic information it contains. It was very nice to have the picture of how this biological process works.
Beyond basic research, what is the implication in biomedicine?
If we know how mitochondrial DNA is packaged and protected from the whole chain of oxidative stress, we can understand what happens in cases where this process is malfunctioning. If the DNA is not well packaged it can be damaged and it has been seen that these lesions can be related to neurodegenerative diseases such as Alzheimer’s and Parkinson’s.
And how you got Heidelberg European Molecular Biology Laboratory (EMBL)?
During the PhD, I visited another institute, the German Cancer Research Center (DKFZ), which is located in Heidelberg. I already knew the city and some people from the EMBL. When I was looking for a postdoc and I received a job offer from Dr. Orsolya Barabas at EMBL. The project was about demethylation of DNA and how it is related to epigenetics. One month after I started working on it, another group published the results. That is why we rescued from a drawer the project in which I have been working for the last three and a half years.
From a drawer?
Yes, it was a project in a very early phase. I wasn’t convinced of working with a protein of bacterial origin and at the beginning it was a little desperate. It is difficult to carry out a project with so many open questions. But we have succeeded.
And what results have you obtained?
We have been able to determine the molecular basis of a mechanism for the expansion of multi-resistance to antibiotics in bacteria by determining the three-dimensional structure and realizing in vitro and in vivo functional studies of a transposase. Transposons are genetic elements that “jump” from one DNA to another and transpose is the protein that allows this DNA copy-paste process. We specifically studied a bacterial transposon that contains resistance to Vancomycin, an antibiotic that is used ultimately when all others have failed.
How important is multi-resistance to antibiotics?
It seems that in 10 years the main medical problem will be multi-resistant bacteria. Our results have led us to find a mechanism that blocks this transfer. In this sense we have designed a peptide, a short sequence of amino acids, which blocks a transposase tail essential for its function. On the other hand, we have also designed a small fragment of DNA that binds specifically to a determinant nucleotide and prevents insertion into target DNA. They are two potential ways to block the activity and, therefore, stop the expansion of antibiotic resistance through this mechanism.
The determination of transposase 3D structure was by means of x-ray crystallography?
Yes. The protein-DNA complexes are always very complicated to crystallize. To obtain a homogeneous sample we created an inactive transposase by mutating an amino acid to have the picture of the mechanism always in the same state. The other variable we played with was the length of the DNA until we found the optimum to form the crystals. To diffract the crystals and get the structure, I have made many trips to the Hamburg synchrotron carrying some dewars.
Dewars?
They are liquid nitrogen containers where we carry the crystals. The travel dewars usually weigh about 15 kilos.
How did you carry them?
One in my backpack and two others, one in each hand. My personal belongings were fitted in free little spaces of the backpack, it forced me to take a few (laughs).
To be in a drawer, the project has proven to be very interesting.
I wanted to publish the results, but I never, nor anyone, thought that this work would be published on Cell. Nowadays, funding is subject to the level of publications and the scientific community has this objective. But scientists should want to tell stories, understand mechanisms and not just focus on publishing great articles.
The scientific community has a lot of pressure and you have to make many sacrifices to achieve results like yours…
In fact, when the referees sent us corrections and comments on the article I was in Chilean Patagonia on vacation. I made an express journey back: 3 hours in bus to Punta Arenas, 4 hours flight to Santiago de Chile, 8 hours flight to Madrid and 1 hour flight in Barcelona. I slept few hours and at 6:00 in the morning I flew to Frankfurt where I took the last bus to Heidelberg, one hour more. At 10 am I was at the laboratory to start working on corrections. In one day I crossed half the world, “pim-pam”.
And now, what are you doing?
Well, at the moment I am thinking about what to do and meanwhile I continue my training in different aspects. The other day, I gave a talk on scientific communication within a Laboratory Technician Training. When I asked what scientific journals they knew about, they answered me: “National Geographic”.
Almost just like Cell …