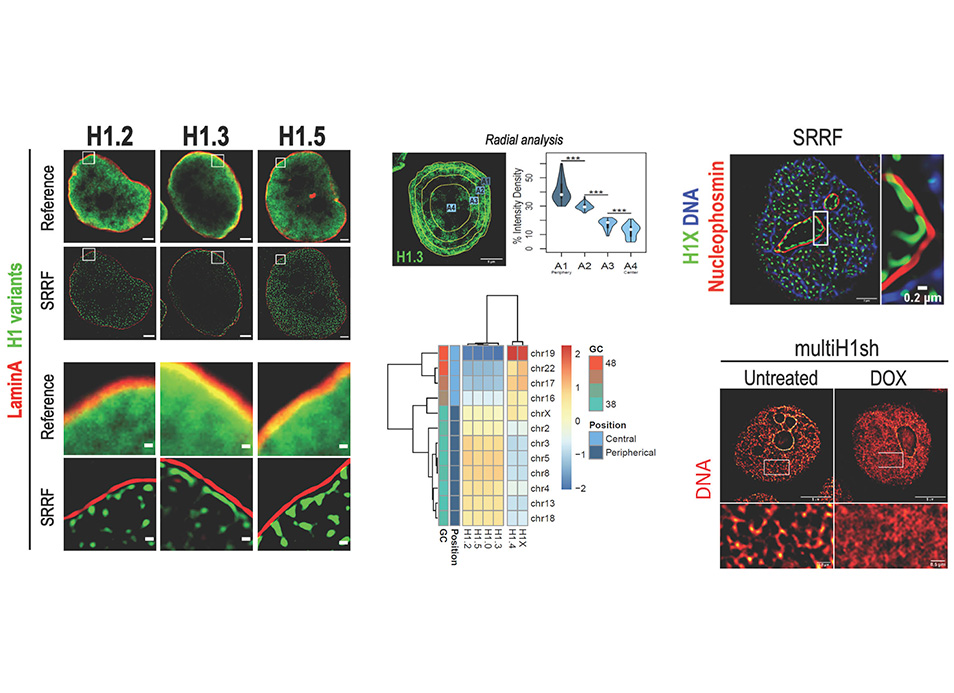
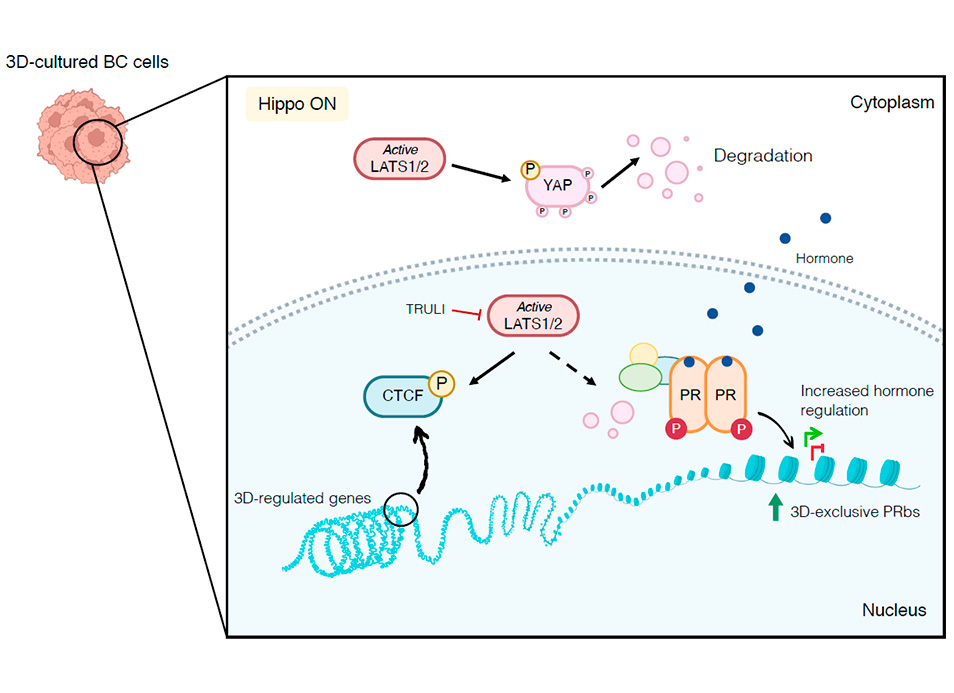
New publication in eLife of the Jordan Lab in collaboration with the IBMB Imaging Platform showing that…
Intermolecular latency regulates the essential C-terminal signal peptidase and sortase of the Porphyromonas gingivalis type-IX secretion system
Intermolecular latency regulates the essential C-terminal signal peptidase and sortase of the Porphyromonas gingivalis type-IX secretion system
A team led by scientists from the Molecular Biology Institute of Barcelona, Higher Council for Scientific Research (IBMB-CSIC) has deciphered an attack mechanism of the principal bacterium that causes severe periodontitis, a disease that affects the gums. The study, published in the PNAS journal, could be used to develop specific oral antiseptics that can kill this bacterium.
The human oral microbiome is made up of more than 6,000 million bacteria from more than 770 species, which are mostly commensal or mutualistic and contribute to the maintenance of homeostasis and good oral health. However, an inadequate diet and insufficient hygiene can lead to alterations in the composition of the microbiome and the excessive growth of pathogenic and opportunistic species, which outnumber the beneficial species and take control of the oral microbiome. This in turn leads to tooth decay and periodontitis, which in the most severe cases leads to chronic inflammation and tissue destruction.
It is estimated that between 5% and 20% of adults and up to 40% of the elderly in Europe suffer from periodontitis, according to WHO figures, and that about 30% of the European population between 65 and 74 years has lost all his teeth from this cause.
Proteins participate in the attack.
Researchers have discovered a key element of the virulence factor secretion system of the bacterium Porphyromonas gingivalis, a pathogenic species of the human oral microbiome. This bacterium has a secretion system (called T9SS) to send proteins (known in scientific language as cargo proteins) to the environment that surrounds it, the gingival tissue that protects the teeth.
These proteins produced by P. gingivalis are involved in bacterial virulence, gum inflammation and tissue destruction, and ultimately, tooth loss in severe periodontitis. The secretion system studied by the scientists, T9SS, has a key enzyme, called PorU, which performs two essential steps so that the cargo proteins can be secreted in a beneficial way for the bacteria.
In collaboration with the Jaguelónica University of Krakow (Poland), the University of Thessaly (Greece) and the University of Louisville (USA), scientists from the Department of Structural Biology of the IBMB-CSIC have solved the structure of the PorU enzyme and have revealed its mechanism of action.
Francesc Xavier Gomis-Rüth, a researcher at the IBMB-CSIC and one of the leaders of the work, explains: “We have been able to determine that a dimerization strategy, a type of chemical reaction, of the PorU protein enzyme is responsible for regulating its activity, repressing it until it reaches the outer membrane of the bacterium, where it is associated with an assembly complex that allows the secretion of cargo proteins ”.
Given that the PorU enzyme is only found in bacterial species, knowledge of its structure and function could be used for the development of specific oral antiseptics against P. gingivalis and, thus, against periodontitis.
Press release by Mercè Fernández / CSIC Communication Catalonia
Reference:
Danuta Mizgalska, Theodoros Goulas, Arturo Rodríguez-Banqueri, Florian Veillard, Mariusz Madej, Ewelina Małecka, Katarzyna Szczesniak, Miroslaw Ksiazek, Magda Widziołek, Tibisay Guevara, Ulrich Eckhard, Maria Solà, Jan Potempa, F. Xavier Gomis-Rüth, PNAS.
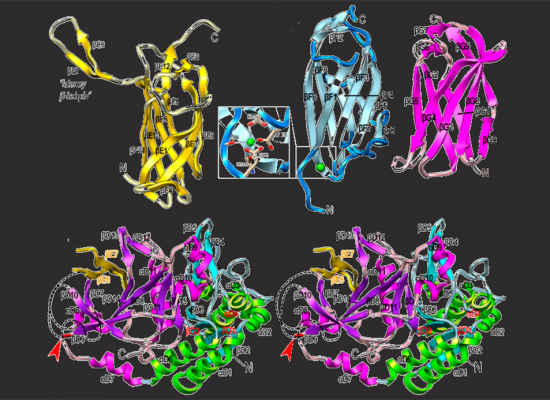
Three-dimensional structure of some of the seven domains of the PorU protein of Porphyromonas gingivalis.